
COMP-Northwest (OR): Steinauer Lab
The main theme that is interwoven throughout all of my scholarly pursuits has been an infectious fascination with the extraordinary success of pathogens. They remain one of the greatest causes of morbidity and mortality worldwide in all living organisms. In humans, pathogens often take a disproportionate toll among those that have the fewest resources and thus these diseases can be notoriously difficult to control. My research seeks to uncover the ecology and evolution of disease in order to inform control efforts and improve health.
Identifying schistosomiasis resistance genes of snail vectors in hotspot transmission zones: Translating from laboratory models to the field
Schistosomiasis is a chronic inflammatory disease that infects over 250 million people, most of whom reside in Africa, and many of whom are children living in poverty [1, 2]. Control of this disease relies primarily on mass drug administration; however, in most endemic transmission foci in Africa, additional approaches are critical if transmission is to be reduced or eliminated. Even after drug treatment, people are rapidly reinfected because the parasites reside and multiply within freshwater snails that are widely distributed in primary water sources. Research efforts to develop mechanisms to block transmission in snails and thus prevent human infection are underway; however, almost all of these efforts have been focused on a South American species of snail, while virtually nothing is known regarding African snail vectors. Our project uses wild populations of African snails to uncover their genetic mechanisms of resistance to infection, so that these mechanisms can be exploited to block schistosomiasis transmission and improve human health. This project is funded through the NIH NIAID R01AI141862

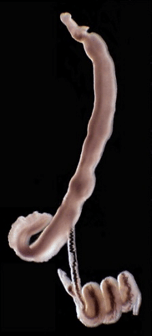
Freshwater snails of the genus Biomphalaria transmit schistosomes to humans. Snails release thousands of parasites daily into the water. Parasites penetrate the skin of humans and cause a chronic inflammatory disease.
The effect of schistosome parasites on host behavior—an interaction through the microbiome?
Schistosomiasis is a chronic disease of inflammation caused by flatworms that invade the vasculature of the intestine or urogenital organs. Chronic infection of school children in endemic countries has been shown to reduce cognitive ability and increase behaviors such as anxiety, hyperactivity, and “acting out” [5]. We hypothesize that schistosomes can influence host behavior through the gut-brain axis indirectly by influencing the microbiome of the intestine. Schistosome eggs passing through the intestinal wall create local inflammation that is likely to influence the microbiome composition and function. In turn, these changes could alter host behavior as inflammation in other disease contexts drives microbiome associated changes in anxiety behaviors and memory in mice.
The primary goal of this project is to determine the effects of schistosome infection on the behavior of a mice as a model of human infection. We have performed a series of infections, behavioral tests, microbiome analysis, along with assays of inflammation and neuropathology to answer this question. Our preliminary results have shown that infection increases anxiety, compulsion, and delayed spatial learning and memory and we are working to determine the underlying mechanisms of these behavior changes.
Two male schistosomes engaged in “tug-of-war” over a female schistosome. Although schistosomes are considered monogamous, mate switching occrus within hosts reflecting competition for mates [3]
Disease Ecology and Evolution
It is becoming increasingly apparent that an individual’s risk of infection and degree of illness is strongly influenced by a variety of ecological factors including abiotic climate factors, coinfecting pathogens, and diversity of host communities. Additionally, because hosts and pathogens can inflict large fitness consequences on their hosts, they have a large role in shaping each other’s evolutionary history and future trajectory. In this framework of thinking, we work on projects involving natural, wild disease systems and include their inherent complexity as well as set up well controlled laboratory experiments to determine the effects of parasitism on life history evolution. It is these approaches together that will help us understand the ecology and evolution of disease processes.

Schistosomes are one of the many pathogens that infect African Buffalo. Climate, nutrition, immune response, and other pathogens all influence the prevalence of schistosomes in buffalo [4]
Citations
Lo NC, Addiss DG, Hotez PJ, King CH, Stothard JR, Evans DS, et al. A call to strengthen the global strategy against schistosomiasis and soil-transmitted helminthiasis: the time is now. Lancet Infect Dis. 2017;17(2):E64-E9. PubMed PMID: WOS:000412365900005.
Steinmann P, Keiser J, Bos R, Tanner M, Utzinger J. Schistosomiasis and water resources development: systematic review, meta-analysis, and estimates of people at risk. Lancet Infect Dis. 2006;6(7):411-25. PubMed PMID: ISI:000238712700023.
Steinauer ML. The sex lives of parasites: Investigating the mating system and mechanisms of sexual selection of the human pathogen Schistosoma mansoni. Int J Parasitol. 2009;39:1157-63.
Beechler BR, Jolles AE, Budischak S, Corstjens PLAM, Ezenwa VO, Smith M, et al. Host immunity, nutrition and coinfection alter longitudinal infection patterns of schistosomes in a free ranging African buffalo population PLoS Neglected Tropical Diseases. 2017;11(12):e0006122.
Musuva R, Shen Y, Wei XJ, Binder S, Ivy JA, Secor WE, et al. Change in children’s school behavior after mass administration of praziquantel for Schistosoma mansoni infection in endemic areas of western Kenya: A pilot study using the Behavioral Assessment System for Children (BASC-2). Plos One. 2017;12(7). PubMed PMID: WOS:000406371800073.
National Library of Medicine – Pubmed Search
M.W. Mutuku, M. R. Laidemitt, B. R. Beechler, I. N. Mwangi, F. O. Otiato, E. L. Agola, H. Ochanda , B. Kamel, M. L. Steinauer, E. S. Loker, G. M. Mkoji. 2019. A search for snail-related answers to explain hotspots of Schistosoma mansoni transmission among villages along the Kenyan shore of Lake Victoria: is the entire lake really a hotspot? American Journal of Tropical Medicine and Hygiene 101(1): 65-77.
Steinauer, M.L., Flores, V., & Rauque, C.A Centrorhynchus rufipus n. sp. (Acanthocephala: Centrorhynchidae) from rufous-legged owl (Strix rufipes King, 1827) in Patagonia. 2019. Journal of Helminthology 94: e42.
Steinauer, M.L., Garcia-Vedrenne, A.E. Weinstein, S.B., & Kuris, A.M. 2019. Acanthocephalan parasites of the oarfish, Regalecus russelii (Regalecidae), with a description of a new species of Gymnorhadinorhynchus (Acanthocephala: Gymnorhadinorhynchidae). Journal of Parasitology 105(1).
Beechler, B.R., A.E. Jolles, Budischak, S., Corstjens, P.L.A.M., Ezenwa, V. O., Smith, M., Spaan, R., van Dam, G.J., & M.L. Steinauer. 2017. Host immunity, nutrition and coinfection alter longitudinal infection patterns of schistosomes in a free ranging African buffalo population. PLoS Neglected Tropical Diseases 11(12): e0006122.
Tavalire, H., M.S. Blouin, & M.L. Steinauer. 2016. Genotypic variation in host response to infection affects parasite reproductive rate. International Journal for Parasitology 46(2): 123-131.
Steinauer, M.L. & B.B. Nickol. 2015. Revision of the Leptorhynchoides thecatus (Acanthocephala: Illiosentidae), with morphometric analysis and description of five new species. Journal of Parasitology 101(2): 193-2011.
Kuris, A.M., A.G. Jaramillo, J.P. McLaughlin, S.B. Weinstein, A.E. Garcia-Vedrenne, G.O. Poinar, Jr., M. Pickering, M.L. Steinauer, M. Espinoza, J.E. Ashford, G.L.P. Dunn. 2015. Monsters of the sea serpent: Parasites of an oarfish Regalecus glesne. Journal of Parasitology 101(1): 41-44.
Lelo, A.E., D.N. Mburu, G.N. Magoma, B.N. Mungai, J.H. Kihara, I.N. Mwangi, G.M. Maina, J.M. Kinuthia, M.W Mutuku, E.S.Loker, G.M. Mkoji& M.L. Steinauer. 2014. No apparent reduction in schistosome burden or genetic diversity following four years of school-based mass drug administration in Mwea central Kenya a heavy transmission area. PLoS Neglected Tropical Diseases. DOI: 10.1371/journal.pntd.0003221.
Steinauer, M.L., M.R. Christie, M.S. Blouin, L.E. Agola, I. N. Mwangi, G.M. Maina, M.W. Mutuku, J.M. Kinuthia, G.M. Mkoji, E.S. Loker. 2013. Non-invasive sampling of schistosome parasites from humans requires correcting for family structure. PLoS Negl Trop Dis 7(9): e2456. doi:10.1371/journal.pntd.0002456.
Steinauer, M.L. & K.M. Bonner. 2012. Host susceptibility is altered by light intensity and photoperiod disruption after exposure to parasites. Journal of Parasitology 2012 98(5): 1052-1054.
Ferguson, J.A., S.A. Locke, W.F. Font, M.L. Steinauer, D.J. Marcogliese, C.D. Cojocaru & M. L. Kent. 2012. Apophallus microsoma n. sp. From chicks infected with metacercariae from coho salmon (Oncorhynchus kisutch) and review of the taxonomy and pathology of the genus Apophallus (Heterophyidae). Journal of Parasitology 98(6): 1122-32.
Cupit, P.M., M.L. Steinauer, B.W. Tonnessen, L.E. Agola, J.M. Kinuthia, I.N. Mwangi, M.W. Mutuku, G.M. Mkoji, E.S. Loker & C. Cunningham. 2011. Polymorphism associated with the Schistosoma mansoni tetraspanin-2 gene. International Journal for Parasitology 41(12): 1249-1252.
Steinauer, M.L., M.S. Blouin, & C.D. Criscione. 2010. Invited review: Applying evolutionary genetics to schistosome epidemiology. Infection Genetics and Evolution 10(4): 433-443.
Hanelt, B., I.N. Mwangi, J.M. Kinuthia, G.M. Maina, L.E. Agola, M.W. Mutuku, M.L. Steinauer, B.R. Agwanda, L. Kigo, B.N. Mungai, E.S. Loker, & G.M. Mkoji. 2009. Schistosomes of small mammals from the Lake Victoria Basin, Kenya: new species, familiar species, and implications for schistosomiasis control. Parasitology 39: 1353-1362.
Steinauer, M.L., B. Hanelt, L.E. Agola, G.M. Mkoji, & E.S. Loker. 2009. Genetic structure of Schistosoma mansoni in western Kenya: the effects of geography and host sharing. International Journal of Parasitology 39: 1353-1362.
Agola, E. L., M.L. Steinauer, D.N. Mburu, B.N. Mungai, I.N. Mwangi, G.N. Magoma, E.S. Loker, & G.M. Mkoji. 2009. Genetic diversity and population structure of Schistosoma mansoni within human infra-populations in Mwea, central Kenya assessed by microsatellite markers. Acta Tropica 111: 219-225.
Hanelt, B., Brant, S.V., Steinauer, M.L., Maina, G.M., Kinuthia, J.M., Agola, L.E., Mwangi, I.N., Mungai, B.N., Mutuku, M.W., Mkoji, G.M., and Loker, E.S. 2009. Schistosoma kisumuensis n. sp. (Digenea: Schistosomatidae) from murid rodents in the Lake Victoria Basin, Kenya and its phylogenetic position among the S. haematobium group. Parasitology 136(2): 211-218.
Melman, S.D., M.L. Steinauer, C. Cunningham, L. Salter-Kubatko, I.N. Mwangi, M. Matuku, D.M.S. Karanja, D.G. Colley, C.L. Black, W.E. Secor, N. Barker, G. Mkoji, & E.S. Loker. 2009. Reduced susceptibility of Kenyan Schistosoma mansoni to praziquantel following repeated exposures: origin, measurement, and likelihood of persistence. PLoS Neglected Tropical Diseases 3(8): e504.
Steinauer, M.L. 2009. The sex lives of parasites: Investigating the mating system and mechanisms of sexual selection of the human pathogen Schistosoma mansoni. International Journal for Parasitology 39: 1157-1163.
Black, C.L., M. L. Steinauer, P.N.M. Mwinzi, W.E. Secor, D.M.S. Karanja, & D.G. Colley. 2009. Impact of intense, longitudinal retreatment with praziquantel on cure rates of schistosomiasis mansoni in a cohort of occupationally exposed adults in western Kenya. Tropical Medicine & International Health 14(4): 1-8.
Hanelt, B., M. L. Steinauer, I.N. Mwangi, G.M. Maina, L.E. Agola, G.M. Mkoji & E.S. Loker. 2009. A new approach to characterize populations of Schistosoma mansoni from humans: development and assessment of microsatellite analysis of pooled miracidia. Tropical Medicine & International Health 14(3): 322-331.
Steinauer, M.L., B. Hanelt, G. Maina, I.N. Mwangi, J. Kinuthia, M. Matuku, G.M. Mkoji, & E.S. Loker. 2008. Hybridization and introgression of human pathogenic schistosomes in Kenya. Molecular Ecology 17: 5062-5074.
Steinauer, M.L., G. Maina, I.N. Mwangi, J. Kinuthia, M. Matuku, G.M. Mkoji, & E.S. Loker. 2008. Interactions between natural populations of human and rodent schistosome parasites in the Lake Victoria region of Kenya: a molecular epidemiological approach. PLoS Neglected Tropical Diseases 2(4): 1-11.
Steinauer, M.L., L. E. Agola, I. N. Mwangi, G. M. Mkoji, & E.S. Loker. 2008. Molecular epidemiology of Schistosoma mansoni: a robust, high-throughput method to assess multiple microsatellite markers from individual miracidia. Infection Genetics and Evolution 8: 68-73.
Steinauer, M.L., B.B. Nickol, & G. Ortí. 2007. Cryptic speciation and patterns of phenotypic variation of a highly variable acanthocephalan parasite. Molecular Ecology 16(19): 4110-4127.
Steinauer M.L., J.E. Parham, & B.B. Nickol. 2006. Geographic analysis of host use, development, and habitat use of an acanthocephalan species, Leptorhynchoides thecatus. Journal of Parasitology 92(3): 464-472.
Steinauer, M.L., B.B. Nickol, R. Broughton, & G. Ortí. 2005. First sequenced mitochondrial genome from the phylum Acanthocephala (Leptorhynchoides thecatus) and its phylogenetic position within metazoa. Journal of Molecular Evolution 60(6): 706-715.
Steinauer, M.L. & W.F. Font. 2003. Seasonal dynamics of the helminths of bluegill (Lepomis macrochirus) in a subtropical region. Journal of Parasitology 89(2): 324-328.
Steinauer, M.L. & B.B. Nickol. 2003. Effect of cystacanth body size on adult success. Journal of Parasitology 89(2): 251-254.
Steinauer, M.L. & B.D. Horne. 2002. The enteric helminths of Graptemys flavimaculata Cagle, 1954 a threatened chelonian species from the Pascagoula River in Mississippi, U.S.A. Comparative Parasitology 69(2): 219-222.
Johannie M. Spaan

I completed a PhD in Zoology at Oregon State University in June 2017, where my work focused on stress physiology in free-ranging female African buffalo. I am currently a postdoctoral fellow in the Steinauer lab at Western University of Health Sciences. Our research focus on Schistosomiasis, a tropical neglected disease that affects over 200 million people. Two main projects that I am involved with are 1) understanding how schistosomes influence the behavior and learning capacity of their hosts, and 2) uncovering potential mechanisms that can be manipulated to break the life cycle of this pathogen in order to reduce or eliminate schistosome transmission to humans. My research interest focus on parasitology, stress physiology, disease ecology, eco-immunology, and bioinformatics.
Reference: Picture taken by P. Buss in August 2012.
One Health Research and Education: Costa Rica

The “One Health” framework stresses the linkages connecting human, animal, and environmental health with the goal of attaining optimal health for all1. This perspective is critical to maintaining health in our current era of extreme anthropogenic change, characterized by climate change, vast urbanization and deforestation, and increased globalization. We are beginning to recognize the profound impacts of “one health” linkages in the context of emerging infectious disease and the global spread of antimicrobial resistance genes. However, one health concepts are difficult to operationalize because they require a collaborative effort across health professions working locally and globally. Although interprofessional education and interprofessional healthcare practice is being recognized as an important step toward improving health care systems2,3, this concept of interprofessional education rarely extends outside of the human health care setting1
 This effort is an integrated research and training program that 1.) provides a platform for cutting-edge, student-led one health research at the wildlife-livestock-human interface in Costa Rica, and 2.) educates future health care professionals in one health principles, research literacy, interprofessional collaboration, and global health. The three main arms of investigation include:
This effort is an integrated research and training program that 1.) provides a platform for cutting-edge, student-led one health research at the wildlife-livestock-human interface in Costa Rica, and 2.) educates future health care professionals in one health principles, research literacy, interprofessional collaboration, and global health. The three main arms of investigation include:
- The spread and flow of antimicrobial resistance genes in domestic animals, wildlife, water supply, and humans.
- The distribution of vectors and their pathogens across a gradient in human land use.
Distribution of water-borne pathogens along a gradient in human land use using environmental DNA.
The Team
Brianna Beechler DVM, PhD, Oregon State University
Rhea Hanselmann, DVM, PhD, Western University of Health Sciences
Anna Jolles PhD, Oregon State University
Elisabeth Guenther MD, MPH, Western University of Health Sciences
Edward Junkins MD, MPH, Western University of Health Sciences
Carrie Manore, PhD, Los Alamos National Laboratory,
Maarten Schrama, PhD, Leiden University
Sofia Solano Costa Rica Conservation Biology
Carlina Fernandez Coghi, DVM and Gianca Coghi, DVM Costa Rica
Citations
- Courtenay, M., Sweeney, J., Zielinska, P., Brown Blake, S. & La Ragione, R. One Health: An opportunity for an interprofessional approach to healthcare. J. Interprof. Care 29, 641–642 (2015).
- Bodenheimer, T., Ghorob, A., Willard-Grace, R. & Grumbach, K. The 10 building blocks of high-performing primary care. Ann. Fam. Med.12, 166–171 (2014).
- Oandasan, I., Baker, G. R. & Barker, K. Teamwork in Health Care: Promoting Effective Teamwork in Healthcare in Canada: Policy Synthesis and Recommendations. (Canadian Health Services Research Foundation, 2006).


