
The Wagner Laboratory focuses on the synaptic and hormonal determinants underlying the central control of energy homeostasis. The research conducted in his lab probes the function of nutrient- and palatability-sensing circuits in the brain, and how dysregulation of these circuits helps establish a link between diet-induced obesity and food addiction.
The Wagner lab focuses on the central regulation of energy balance (i.e., energy intake and expenditure). We probe the influence of sex and the activational effects of gonadal hormones on the signaling of critical neuropeptides (e.g., nociceptin/orphanin FQ (N/OFQ), pituitary adenylyl cyclase-activating polypeptide (PACAP)) and neuromodulators (e.g., endocannabinoids) within the homeostatic (i.e., nutrient sensing) and hedonic (i.e., palatability sensing) energy balance circuits. We also evaluate the impact of energy status across the spectrum – from negative energy balance (e.g., fasting) to positive energy balance (e.g., diet-induced obesity) – on the functioning of these distinct yet overlapping circuits. We use state-of-the-art optogenetic and chemogenetic techniques in strategically determined transgenic animal models to elicit cell type-specific excitation or inhibition of critical neuroanatomical substrates within these circuits and ascertain the corresponding changes in energy intake, meal pattern and energy expenditure. Our work provides direct translational insights into the underpinnings to clinically relevant conditions such as HIV/AIDS-related cachexia and diabetes/insulin resistance.

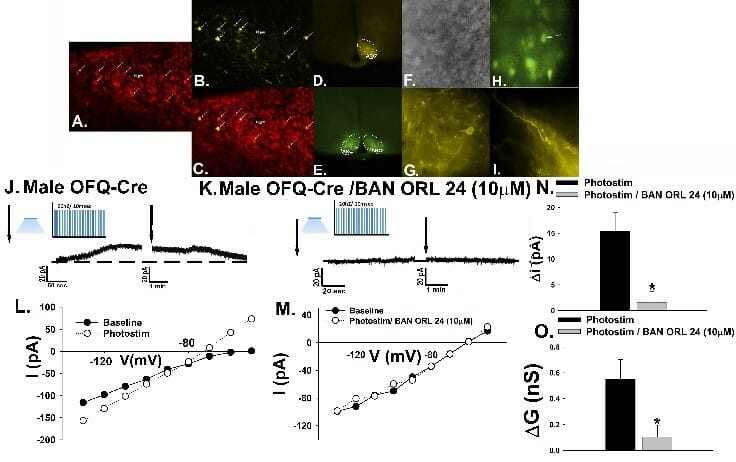
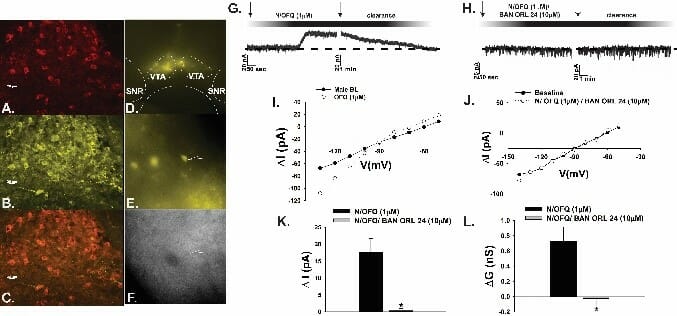
We have shown over the years that N/OFQ profoundly impacts neuronal activity within critical anorexigenic (i.e., appetite suppressing) circuits involved in the hypothalamic control of energy homeostasis. This occurs through both presynaptic and postsynaptic mechanisms. As shown below, optogenetic stimulation of ARC N/OFQ neurons inhibits anorexigenic proopiomelanocortin (POMC) neurons in double transgenic N/OFQ-cre/ eGFP POMC mice. The photomicrograph below illustrates the N/OFQ immunostaining in the ARC visualized with AF546 and colocalized with channelrhodopsin (ChR2; visualized with an eYFP reporter signal. The photostimulation elicits a robust and reversible, nociceptin opioid (NOP) receptor-mediated outward current in the POMC neurons that reverses polarity near the equilibrium potential for K+ and is accompanied by increased slope conductance.
Within the hedonic circuitry, our recent findings demonstrate that N/OFQ also inhibits mesolimbicortical (A10) dopamine neurons in the ventral tegmental area (VTA) that influence natural and drug-induced reward as well as addiction. This occurs upon activation of NOP receptors via the same mechanism as describe above for POMC neurons.
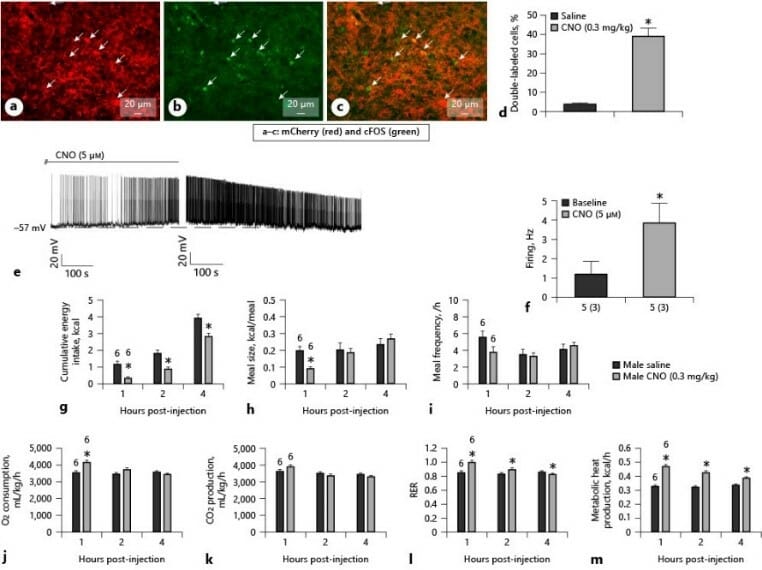
As mentioned above, one of our main research objectives is focused on the critical anorexigenic circuit within the hypothalamic energy balance circuitry comprising upstream PACAP neurons in the ventromedial nucleus that synapse with POMC neurons in the arcuate nucleus. Chemogenetic activation of PACAP neurons increases cFOS expression and the firing rate within these cells. This results in decreased energy intake and meal size, which is accompanied by an increase in energy expenditure.
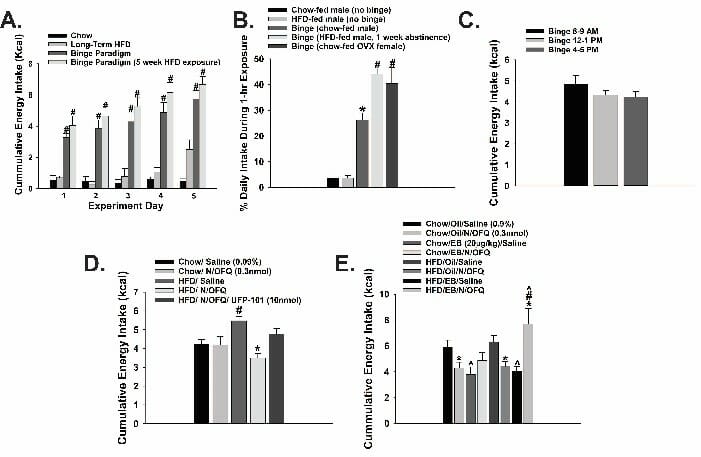
Our current studies regarding N/OFQ are aimed to test the hypothesis that N/OFQ regulates homeostatic energy balance circuitry in a sex- and diet-dependent manner by specifically determining whether:
We also are looking to test the hypothesis that N/OFQ modulates circuits regulating energy balance and related appetitive behavioral processes in a sex- and diet-dependent manner as assessed by the ability of:
• Chang R, Hernandez J, Gastelum C, Guadagno K, Perez L, Wagner E, J: Pituitary Adenylate Cyclase-Activating Polypeptide Excites Proopiomelanocortin Neurons: Implications for the Regulation of Energy Homeostasis. Neuroendocrinology 2020. doi: 10.1159/000506367
• Hernandez J, Fabelo C, Perez L, Moore C, Chang R, Wagner EJ. Nociceptin/orphanin FQ modulates energy homeostasis through inhibition of neurotransmission at VMN SF-1/ARC POMC synapses in a sex- and diet-dependent manner. Biol Sex Differ. 2019;10(1):9. Published 2019 Feb 12. doi:10.1186/s13293-019-0220-3
• Fabelo C, Hernandez J, Chang R, et al. Endocannabinoid Signaling at Hypothalamic Steroidogenic Factor-1/Proopiomelanocortin Synapses Is Sex- and Diet-Sensitive. Front Mol Neurosci. 2018;11:214. Published 2018 Jun 19. doi:10.3389/fnmol.2018.00214
• Qiu J. Bosch MA, Meza C, Navarro U, Nestor CC, Wagner EJ, Rønnekleiv OK and Kelly MJ. Estradiol Protects Proopiomelanocortin Neurons Against Insulin Resistance. Endocrinology. February 2018, 159(2):647-664.
• Qiu J, Wagner EJ, Rønnekleiv OK, Kelly MJ. Insulin and Leptin Excite Anorexigenic Pro-Opiomelanocortin Neurones via Activation of TRPC5 Channels. Journal of Neuroendocrionolgy. June 2017. DOI: 10.1111/jne.12501.
• Conde K, Fabelo C, Krause WC, Propst R, Goethel J, Fischer D, Hur J, Meza C, Ingraham HA, Wagner EJ. Testosterone Rapidly Augments Retrograde Endocannabinoid Signaling in Proopiomelanocortin Neurons to Suppress Glutamatergic Input from Steroidogenic Factor-1 Neurons via Upregulation of Diacylglycerol Lipase-a. Neuroendocrinology. 2016 Nov. 21 PMID:27871072
• Wagner EJ. Sex differences in cannabinoid-regulated biology: A focus on energy homeostasis. Front Neuroendocrinol. 2016 Jan;40:101-9. doi 10.1016/j.yfrbe.2106.01.003. Epub 2016 Jan 19.
• Conde K, Meza C, Kelly MJ, Sinchak K, Wagner EJ. Estradiol Rapidly Attenuates ORL-1 Receptor-Mediated Inhibition of Proopiomelanocortin Neurons via Gq-Coupled, Membrane-Initiated Signaling. Neuroendocrinology. 2016 Jan 16. PMID:26765570
• Mela V, Vargas A, Meza C, Kachani M, Wagner EJ. Modulatory influences of estradiol and other anorexigenic hormones on metabotropic:Gi/O-coupled receptor function in the hypothalamic control of energy homeostasis. J Steroid Biochem Mol Biol. 2015 Jul 28. pii: S0960-0760(15)30027-3. doi:10.1016/j.jsbmb.2015.07.014.
• Borgquist A, Meza C, Wagner EJ. Role of neuronal nitric oxide synthase in the estrogenic attenuation of cannabinoid-induced changes in energy homeostasis. J Neurophysiol. 2015 Feb 1;113(3):904-14. doi: 10.1152/jn.00615.2014. Epub 2014 Nov 12.
• Borgquist A, Meza C, Wagner EJ. The role of AMP-activated protein kinase (AMPK) in the androgenic potentiation of cannabinoid-induced changes in energy homeostasis. Am J Physiol Endocrinol Metab. 2014 Dec 30:ajpendo.00421.2014. doi: 10.1152/ajpendo.00421.2014. https://www.ncbi.nlm.nih.gov/pubmed/25550281
• Qiu J, Zhang C, Borgquist A, Nestor CC, Smith AW, Bosch MA, Ku S, Wagner EJ, Rønnekleiv OK, Kelly MJ. Insulin excites anorexigenic proopiomelanocortin neurons via activation of canonical transient receptor potential channels. Cell Metab. 2014 Apr 1;19(4):682-93. doi: 10.1016/j.cmet.2014.03.004.
• Borgquist A, Kachani M, Tavitian N, Sinchak K, Wagner EJ. Estradiol negatively modulates the pleiotropic actions of orphanin FQ/nociceptin at proopiomelanocortin synapses. Neuroendocrinology. 2013;98(1):60-72. doi: 10.1159/000351868. Epub 2013 Jul 2.
• Sinchak K, Wagner EJ. Estradiol signaling in the regulation of reproduction and energy balance. Front Neuroendocrinol. 2012 Oct;33(4):342-63. doi: 10.1016/j.yfrne.2012.08.004. Epub 2012 Sep 7.
• Viveros MP, Mendrek A, Paus T, Lopez-Rodriguez AB, Marco EM, Yehuda R, Cohen H, Lehrner A, Wagner EJ: A comparative, developmental, and clinical perspective of neurobehavioral sexual dimorphisms. Frontiers in Neuroscience 2012 Jun 12;6:84. doi: 10.3389/fnins.2012.00084.
• Washburn N, Borgquist A, Wang K, Jeffery GS, Kelly MJ, Wagner EJ: Receptor subtypes and signal transduction mechanisms contributing to the estrogenic attenuation of cannabinoid-induced changes in energy homeostasis. Neuroendocrinology 2013;97(2):160-75. doi: 10.1159/000338669.
• Viveros MP, Bermudez-Silva FJ, Lopez-Rodriguez AB and Wagner EJ. The Endocannabinoid System as Pharmacological Target Derived from Its CNS Role in Energy Homeostasis and Reward. Applications in Eating Disorders and Addiction. Pharmaceuticals 2011, 4(8), 1101-1136; doi:10.3390/ph4081101.
• Viveros MP, Marco E, Lopez-Gallardo M, Garcia-Segura LM and Wagner EJ. Framework for sex differences in adolescent neurobiology: A focus on cannabinoids. Neuroscience and Biobehavioral Reviews 2011, 35 (8), pp. 1740-1751.
• Jeffery GS, Peng KC and Wagner EJ. The role of phosphatidylinositol-3-kinase and AMP-activated kinase in the rapid estrogenic attenuation of cannabinoid-induced changes in energy homeostasis. Pharmaceuticals 2011, 4 (4), pp. 630-651.
Jennifer Hernandez received her Bachelor of Science degree in Biology from California State University, Los Angeles in 2016. She then earned a Masters of Science degree in Biomedical Sciences from Western University of Health Sciences in 2018. She did her master’s thesis work under the supervision of Dr. Ed Wagner at Western University of Health Sciences and was hired as a full time research associate for the same lab in the summer of 2018. During this time she became proficient in electrophysiological recordings from a particular subset of neurons involved in the hypothalamic feeding circuity. She was also instrumental in establishing colonies of transgenic mice including eGFP-POMC, NR5A1-Cre, eGFP-POMC/NR5A1-Cre, and PACAP-Cre. She not only established the colonies, but has successfully implemented and developed the genotyping procedures for these different strains. During her research, she has also become proficient at different surgical procedures such as stereotaxic viral injections and cannulations, as well as ovariectomies. She has also become well versed in iv vivo feeding and metabolic studies for both mice as well as guinea pig models and has performed numerous in vitro studies involving the aforementioned strains. These different procedures have expanded her knowledge of the hypothalamic feeding circuitry, as well as the sex differences and dietary differences that affect energy balance. She has also played a role in mentoring and teaching various masters, medical, veterinary, and high school students during her time at Western University.